How it works
Your QMS, built in seconds.
No configuration. No consultants. No days of setup. Paste in what you already have, click go, and walk away with a complete, audit-ready QMS.
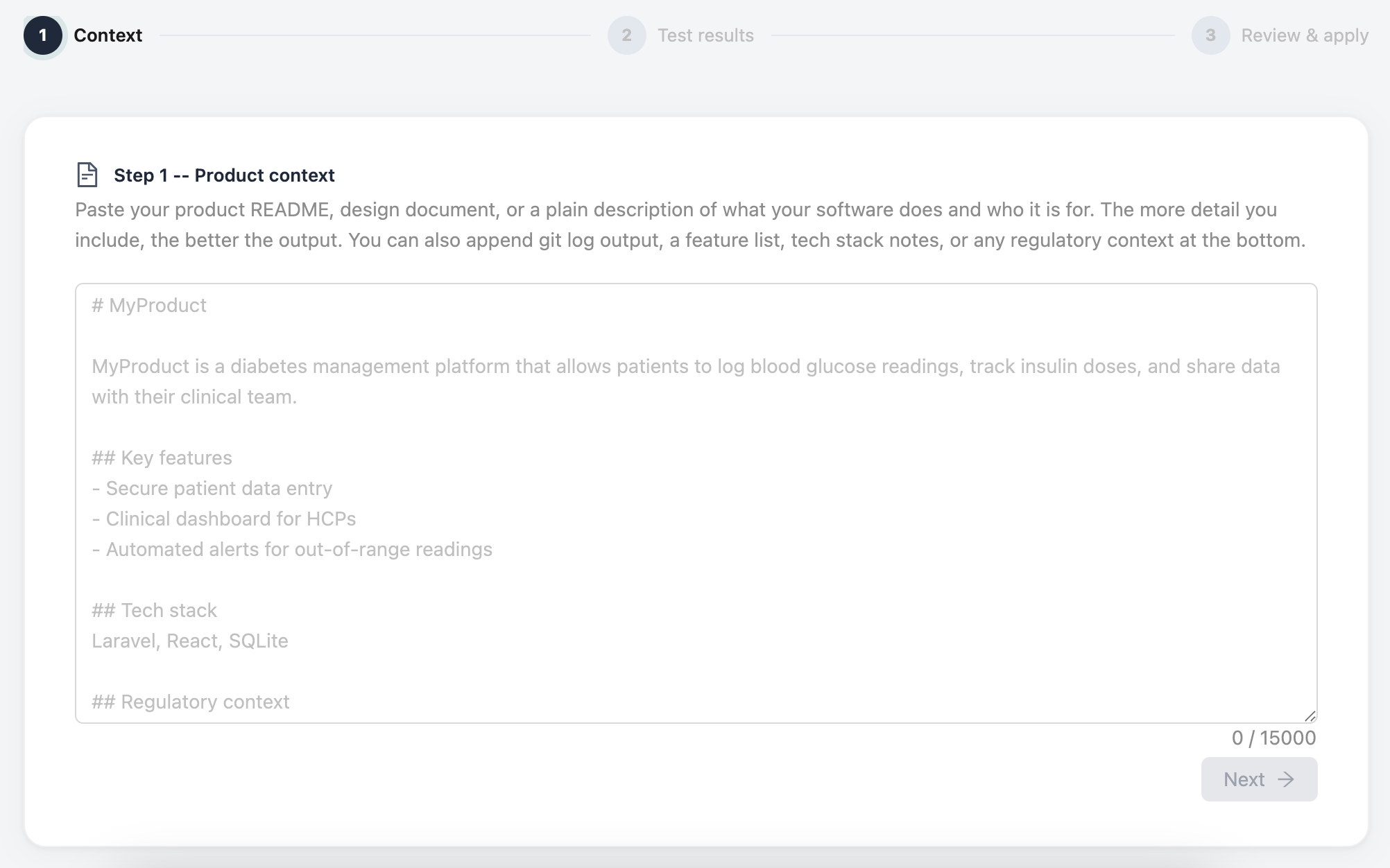
Describe your product
Paste in your README, product spec, or a plain description of what your software does and who uses it. Include your tech stack and any regulatory context you have. That's all QualiHQ needs.
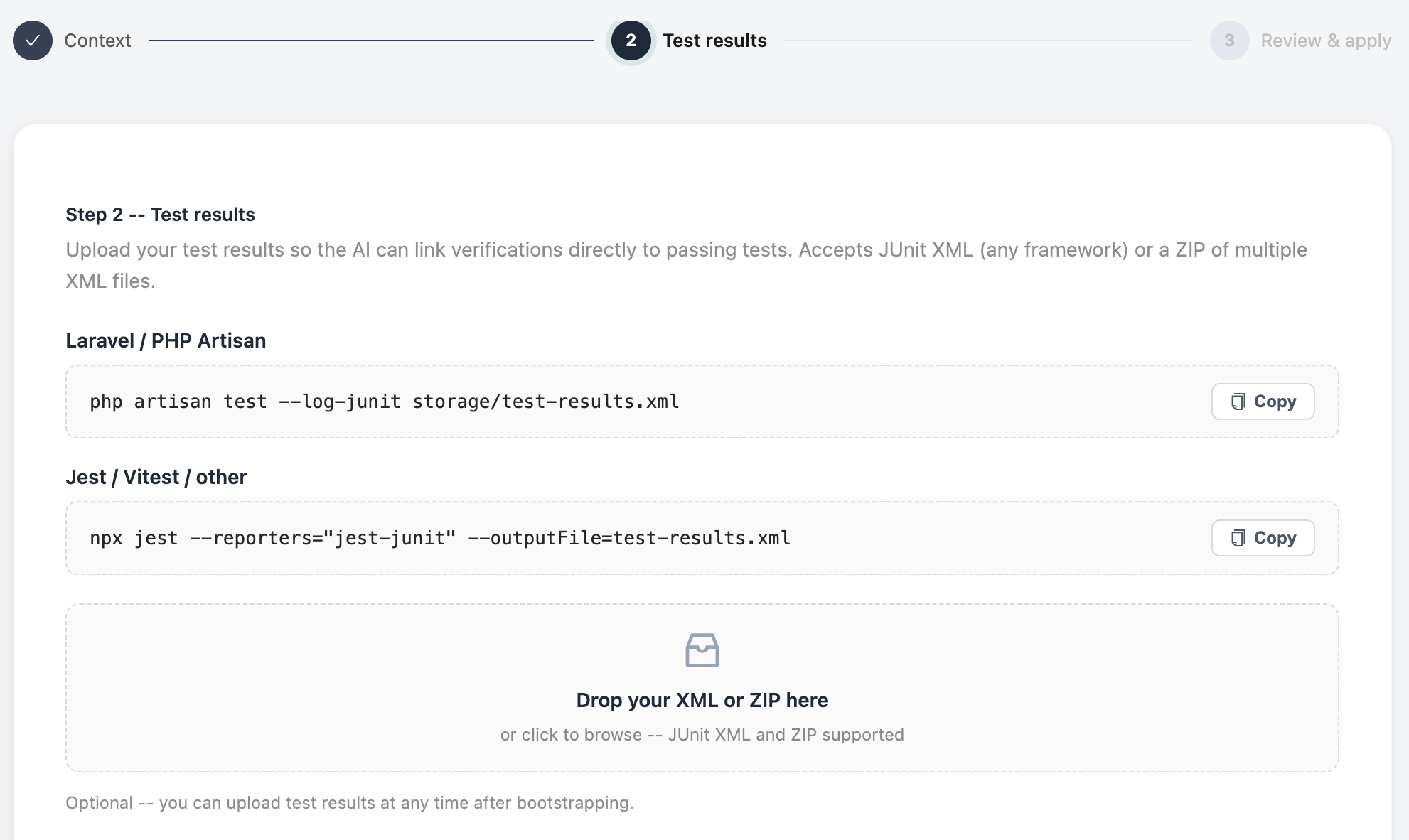
Add your test results and click Go
Paste or upload your test output -- JUnit XML, JSON, or plain results from your CI pipeline. Hit Go and QualiHQ's AI builds your full QMS structure: requirements, verifications, risk analysis, and a complete traceability matrix, all linked and ready to review.
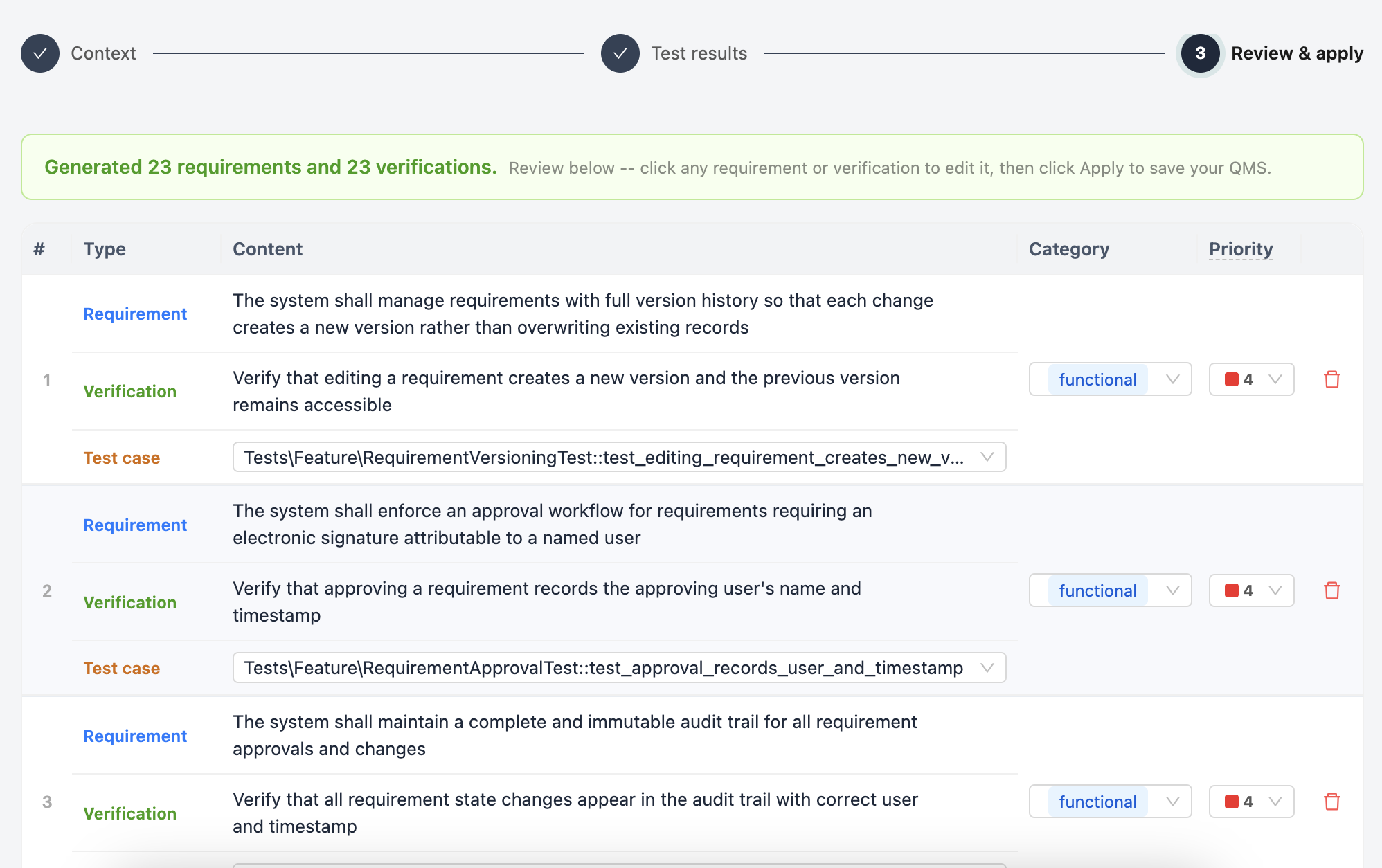
Review, approve, and ship
Everything QualiHQ AI generates lands in a review queue. Go through each requirement, verification, and risk item -- approve what's right, adjust what isn't. Nothing goes live until a human signs off. Once approved, your QMS is complete, your audit trail is built, and you are ready to release.
Everything included
Built for the full compliance lifecycle
Traceability Matrix
Every requirement is automatically linked to its verification and test evidence. Auditors get a complete, navigable traceability matrix with no manual work.
Release Approval Workflow
Structured release management with required reviews and sign-offs. Releases are blocked until all requirements have passing verification evidence and the risk assessment has been reviewed.
Audit Trail
Every action is logged with timestamp, user, and context. Your audit trail is always current, structured for 21 CFR Part 11 requirements, and exportable.
Issue and CAPA Tracking
Log non-conformances, investigate root causes, and manage corrective and preventive actions. Every issue is linked back to the requirements and verifications it affects.
Team and Role Management
Invite your QA leads, engineers, and regulatory affairs team. Role-based access controls ensure the right people can approve, edit, or view each part of your QMS.
Standards Coverage
Built for teams working towards ISO 13485, IEC 62304, and 21 CFR Part 11. QualiHQ structures your QMS around the requirements these standards expect.
Ready in seconds, not months
Start free. Early adopter pricing locked in for the first 50 customers.
Request access →